|
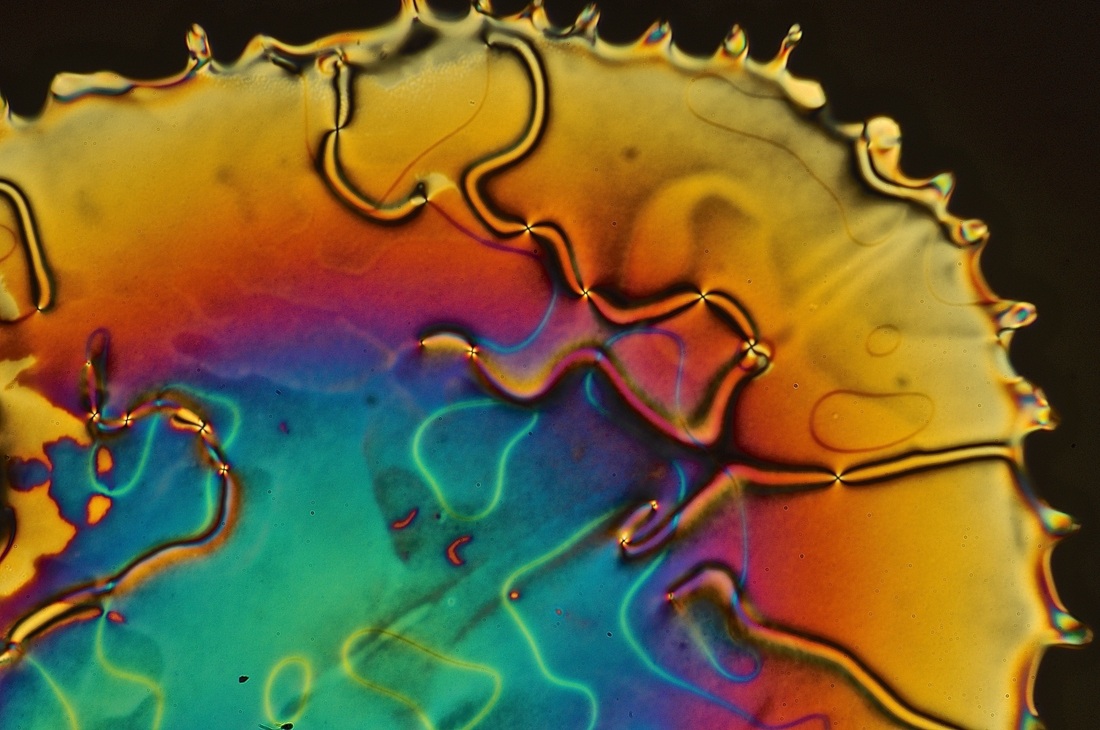
It occurred to me as I was starting this blog that there are a lot of specialized terms in our field. My initial thought was to create blog entry giving definitions of all the terms that I could think of. But who, really, wants to read that? So I'm taking a different tack; I'll introduce the terms one at a time, with lots of pictures. Let's start with the most obvious one term. What are liquid crystals? Liquid crystals are ordered liquids. Ordinary liquids (water, gasoline, oil, etc.) are disordered at the molecular level. Liquid crystals, on the other hand, are made up of molecules that display some form of order. Molecular ordering is a characteristic usually associated with crystalline solids (e.g. sugar and salt). The main difference between liquid crystals and crystalline solids is their degree of ordering; the molecules of liquid crystal phases are not held in a perfect 3D matrix, as is the case for most solids. They have wiggle room. They can flow. That's the liquid part. Ordered fluids have unique advantages, not the least of which is that they can be used to make LCDs (which stands for "liquid crystal display"). I will return to LCDs at some later date. One of their interesting features is that they give pretty textures when viewed by POM. In contrast, ordinary "isotropic" liquids appear completely black in polarized microscopy.
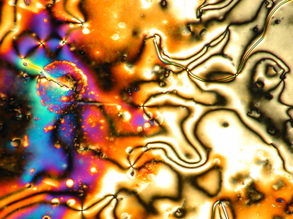
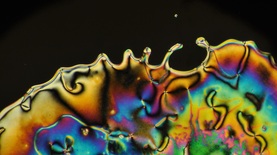
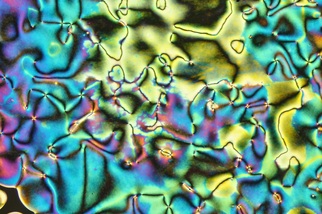
There are some caveats to this generalization that we'll explore more fully in subsequent posts. For example, the black wavy lines that snake their way though the LC phase are not actually isotropic regions. These "disclination lines" are a common feature of many liquid crystals, especially nematic phases. For example: I'll leave you with one last image that nicely shows the fluid nature of the nematic phase; here, the rivulets of liquid crystal extend into the region of ordinary liquid:
Every so often, I get an email from someone who wants to use one of my liquid crystal photographs for some purpose or another. Once, this was for a book cover; another time, it was for a presentation. Yesterday, I was asked for permission to use a liquid crystal picture for the cover of a Ph.D. thesis. Curious about which picture it was, I followed the link to a long defunct version of my SFU webpage. To put it mildly, this page had not aged well: to see it in all of its retro-glory, click here. Here's an important lesson: nothing ever disappears from the internet.
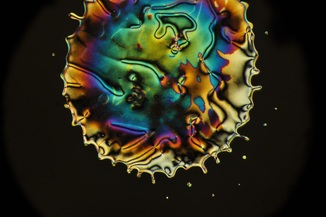
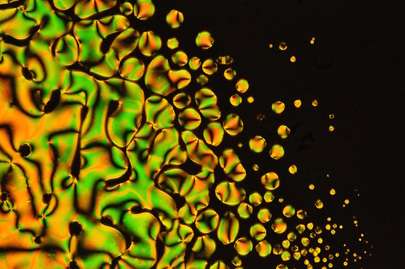
I have a soft spot for nematic phases, for a variety of reasons that I'll explore in later posts. One of these reasons is that nematic phases were the first kind of liquid crystal that I worked with as a grad student. So, for all of those researchers out there who worked with PCH5, here are a few more recent pictures.
Welcome to the LC Texture Blog. I started this blog with two main goals:
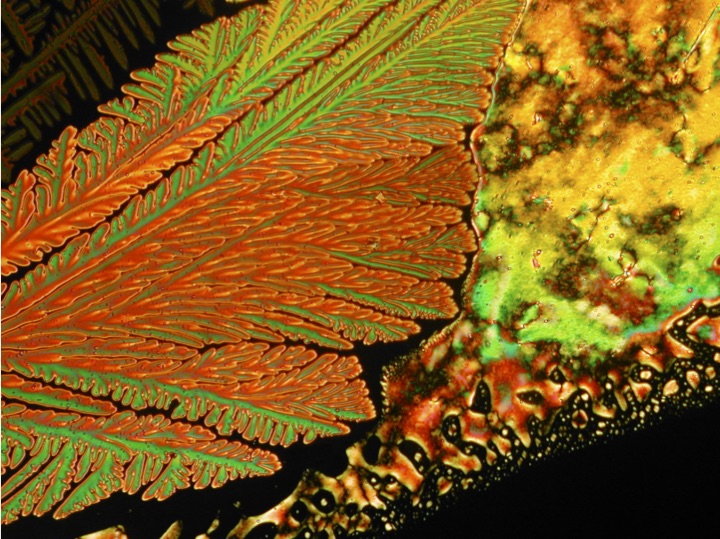
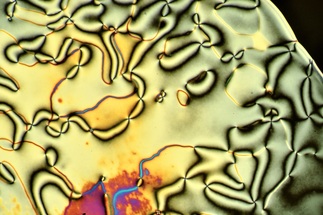
1) to showcase off the beauty of liquid crystals, and 2) provide a forum for discussing the art and science of liquid crystal optical microscopy. Hopefully the aesthetics of liquid crystals will be self-explanatory. The second bit needs a bit of background. Polarized optical microscopy (or "POM") is the go-to technique for liquid crystal researchers. An expert practitioner can often infer the underlying molecular order of a liquid crystal by examining the characteristic optical patterns (or "textures") the material exhibits under a microscope. So for a liquid crystal researcher, these pictures aren't just pretty, but also provide crucial information. This is why we often refer to the optical microscopy of liquid crystals as textural analysis. I am far from an expert at textural analysis. However, over the course of two decades of working with liquid crystals, I have managed to pick up some of the tricks of the trade. My goal is to pass on some of these tricks to newcomers to the field. And if you're not a liquid crystal researcher, then just enjoy the pretty pictures. Like this one of a nematic liquid crystal: |
Vance Williams
is a Professor of Chemistry at Simon Fraser University. Archives
October 2015
Categories |






 RSS Feed
RSS Feed
