|
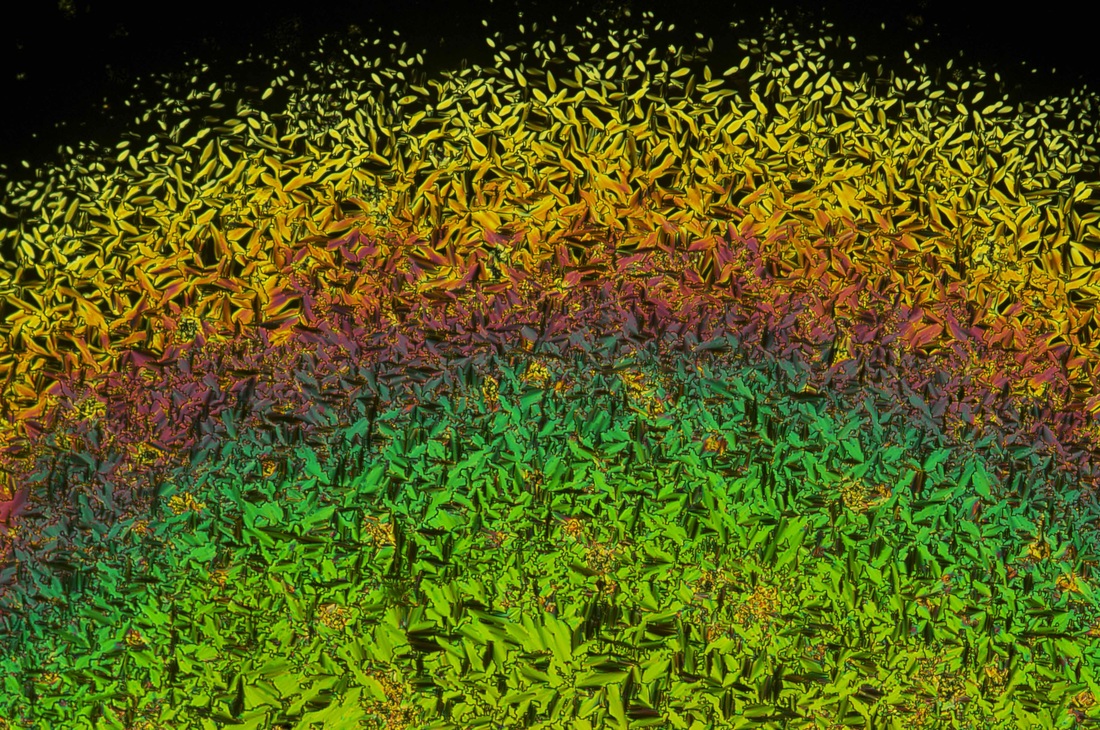
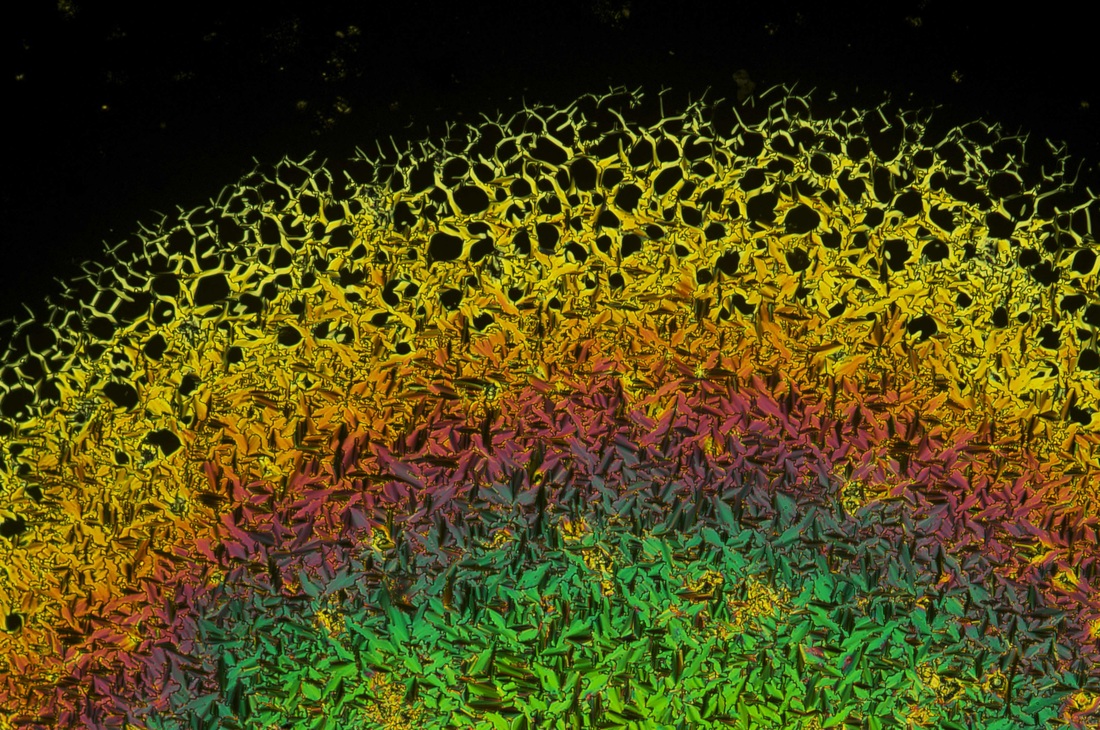
Here's today's fun fact: the majority of organic solids are birefringent. Take, for example, urea:
I took a couple weeks off from blogging as I was getting caught up on other things. I have also started a new side project for myself: microscopy of natural product solids. This falls under the heading of "chemistry outreach." Finding ways to engage the wider public about chemistry is always challenging, so lately I've been taking a different approach: posting pictures of compounds like caffeine, cholesterol, etc. I'll post some of these here in the days to come. For those who just want to look at liquid crystals, don't worry, I'll keep posting those too. For now, here's some morning caffeine:
Chiral nematic phases were the first liquid crystals that I worked with as an undergrad. My Ph.D. was all about these twisted phases. The following movie shows the dramatic effects in texture seen at the clearing temperature when air currents play across the surface of the sample. The sample is a mixture of cholesteryl propionate and 6,6'-bis(hexyloxy)azoxybenzene. |
Vance Williams
is a Professor of Chemistry at Simon Fraser University. Archives
October 2015
Categories |









 RSS Feed
RSS Feed
