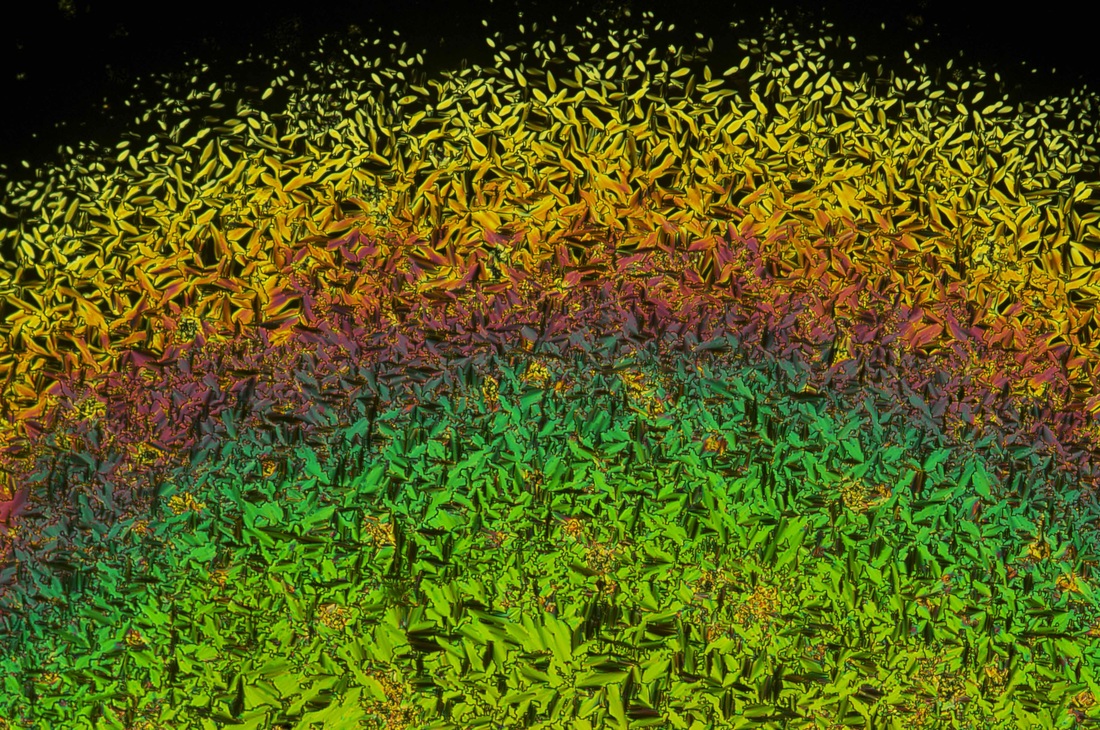
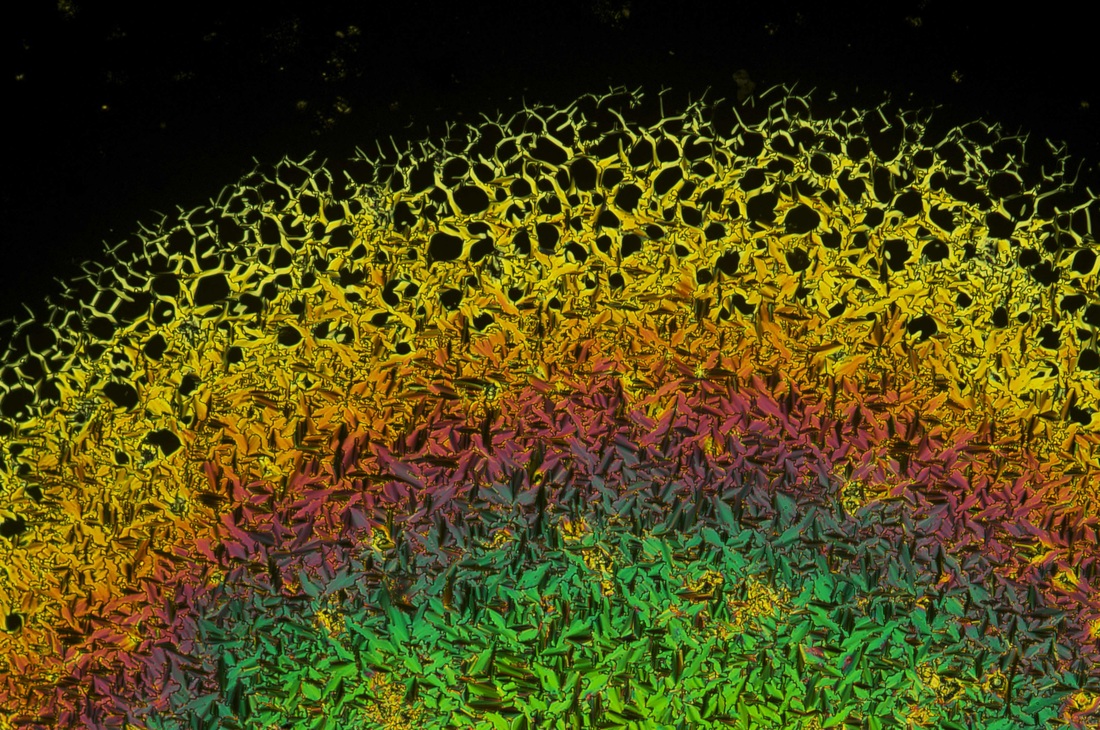
I took a couple weeks off from blogging as I was getting caught up on other things. I have also started a new side project for myself: microscopy of natural product solids. This falls under the heading of "chemistry outreach." Finding ways to engage the wider public about chemistry is always challenging, so lately I've been taking a different approach: posting pictures of compounds like caffeine, cholesterol, etc. I'll post some of these here in the days to come. For those who just want to look at liquid crystals, don't worry, I'll keep posting those too. For now, here's some morning caffeine:
Chiral nematic phases were the first liquid crystals that I worked with as an undergrad. My Ph.D. was all about these twisted phases. The following movie shows the dramatic effects in texture seen at the clearing temperature when air currents play across the surface of the sample. The sample is a mixture of cholesteryl propionate and 6,6'-bis(hexyloxy)azoxybenzene. When liquid crystals were discovered way back in 1888, the molecules being examined were cholesterol derivatives. These phases were dubbed "cholesteric" liquid crystals, a name that has largely stuck. We now know that these are chiral nematic, denoted N* phases. The "*" is used in the field to denote chiral phases. The movie shown below is an attempt to capture the phase transition of an N* phase. For this experiment, the temperature was held at the clearing temperature; the LC and isotropic domains grow and shrink, largely because of air currents in the lab. The last movie that I posted was of a columnar hexagonal phase. As I noted in that post, these phases are often not all that visually interesting, especially compared to nematic phases. This is partly due to symmetry and alignment effects; in a homeotropic orientation, the hexagonal phase is nonbirefringent. In contrast, columnar oblique phases are highly birefringent. The following movie shows an oblique phase growing in from the higher temperature nematic phase. This transition has been featured in earlier photos on this blog, but the movie tells a more dramatic picture: |
Vance Williams
is a Professor of Chemistry at Simon Fraser University. Archives
October 2015
Categories |




 RSS Feed
RSS Feed
